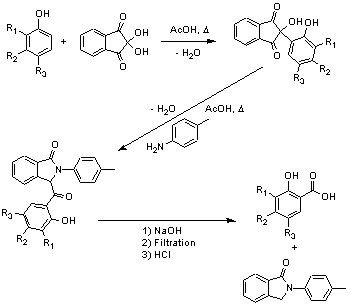
Condensation of phenols with ninhydrin; Substituted salicylic acids SyntheticPage 6 (2001)
Submitted 11th Jun 2001, published 11th Jun 2001
Ian Munslow
([email protected]),
A contribution from the Peter Scott
Chemicals Used
phenol (1 equiv.), ninhydrin (1 equiv.), 4-methylaninine (2 equiv.), acetic acid (100 ml / 0.1 mol), 2N sodium hydroxide (30 ml / 0.05 mol), H2O (10 ml / 0.05 mol), 6N hydrochloric acid (10 ml / 0.05 mol)
Procedure
The phenol is added to a hot solution of ninhydrin in acetic acid and the mixture heated at reflux for 2h. After cooling to room temperature the resulting precipitate was isolated by vacuum filtration (1), washed with ice-cold acetic acid and dried in vacuo, 2nd crop can be collected.
4-Methylaniline is added to a solution of (1) in acetic acid and stirred under reflux for 12h The mixture is allowed to cool to room temperature before the resulting precipitate was isolated by vacuum filtration, washed with ice-cold acetic acid and dried in vacuo, 2nd crop can be collected. (2)
(2) is added to aqueous 2N NaOH and refluxed for 15 min, resulting white ppt was isolated by filtratrion and washed with water. Yellow filtrate and washing were then cooled and acidified with 6N HCl, resulting white ptp is isolated by vacuum filtration, washed with cold water and recrystallized from ethanol/water to give a white solid.
Author's Comments
Procedure works well on the large scale, several grams of pure salicyclic acid being available in a day or two. Each step has crystalline end products, no purification is needed.
Compounds made: 3-iso-propyl-2-hydroxybenzoic acid, 3,5-di-tert-butyl-2-hydroxybenzoic acid, 3,5-di-methyl-2-hydroxybenzoic acid.
Data
Depends on the starting phenol.
3-iso-Propyl-2-hydroxybenzoic acid
1H NMR (CDCl3): 10.91 (brs, 1H, OH), 7.82 (d, J = 6, 1H, Ar-H), 7.45 (d, J = 6, 1H, Ar-H), 6.88 (t, J = 6, m1H, Ar-H), 4.71 (brs, 1H, OH), 3.38 (sept, J = 7, 1H, CHMe2), 1.23 (d, J = 7, 6H, CHMe2).
Lead Reference
Schmitt, G. Synthesis 1984, 758.
Other References
This page has been viewed approximately 338 times since records began.
Get CDX file (Choose "Open this file from its current location" TWICE)
SyntheticPagesTM are for use
exclusively by those with training and experience in synthetic chemistry. While contributors
are asked to identify hazards and to use methods designed to reduce risk, no formal hazard
or risk assessments are included, and these procedures must be conducted at one's own risk.
SyntheticPages, its editors, owners and associates do not warrant or guarantee the safety of
individuals using these procedures and hereby disclaim any liability for any injuries or damages
claimed to have resulted from or related in any way to the procedures herein. Copyright ©2005 Synthetic Pages
Comment on this page!
Registered users may add comments to other pages. If you have already registered, please
log in, otherwise you may
register here.Back to List
|