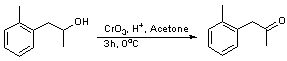Jones oxidation of 1-o-tolyl-propan-2-ol; 1-o-tolyl-propan-2-one SyntheticPage 171 (2001)
Submitted 22nd Nov 2001, published 22nd Nov 2001
lisa frost
([email protected]),
A contribution from the Caddick Group, Sussex
Chemicals Used
Chromium trioxide (Lancaster)
1-o-tolyl-propan-2-ol
Acetone (GPR)
conc H2SO4 (BDH)
water
Procedure
Chromium trioxide (12.8 g, 0.128 mol) was dissolved in 18 mL water (142 mL/molCrO3)in a beaker. The resulting solution was cooled to 0oC whereupon 11 mL conc H2SO4 (87 mL/mol CrO3) was added dropwise. The solution was diluted with 36 mL water (285 mL/mol Cr03) and cooled to 0oC. A solution of the alcohol (19.2 g, 0.128 mol) in acetone (80 mL) (600mL/mol CrO3) was cooled to 0oC and the Jones reagent added slowly, maintaining a temperature <20oC. Stirring was continued for 3 h at 0oC. Sodium bisulfite was added in small portions until the brown colour of the chromic acid had disappeared from the upper layer. The top layer was decanted and the lower layer extracted with ether (2 x 200 mL). The extracts were combined and washed with NaCl solution, dried over MgSO4, filtered and concentrated in vacuo. The resulting brown oil was purified by chromatography on silica gel with petrol:ethyl acetate (10:1) as the eluent yielding the title compound as a yellow oil (14.2g, 75%).
Author's Comments
Although this reaction is very commonly used in synthesis, I found it difficult to find exact conditions when searching. The literature assumes that everyone knows how to make the reagent! Although I have only carried out this reaction once, I found it high yielding and easy to follow.
Data
1H nmr (300 MHz, CDCl3)2.15 (3H, s), 2.26 (3H, s), 3.72 (2H, s), 7.13-7.20 (4H, m).
Lead Reference
Shibuya, Masayuki; Tetrahedron Lett.; 1983; 1175-1178
Other References
Org Synth Coll. 5, 1973, 866
This page has been viewed approximately 903 times since records began.
Get CDX file
SyntheticPagesTM are for use
exclusively by those with training and experience in synthetic chemistry. While contributors
are asked to identify hazards and to use methods designed to reduce risk, no formal hazard
or risk assessments are included, and these procedures must be conducted at one's own risk.
SyntheticPages, its editors, owners and associates do not warrant or guarantee the safety of
individuals using these procedures and hereby disclaim any liability for any injuries or damages
claimed to have resulted from or related in any way to the procedures herein. Copyright ©2005 Synthetic Pages
Comment on this page!
Registered users may add comments to other pages. If you have already registered, please
log in, otherwise you may
register here.Back to List
|