 |
||
 | ||
 |
NBS allylic bromination of cyclohexene; 3-Bromocyclohexene SyntheticPage 144 (2001)Submitted 28th Aug 2001, published 28th Aug 2001 Andrew McCarroll
([email protected]), 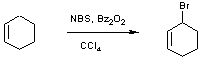
Chemicals Used Procedure Author's Comments Data Lead Reference Other References This page has been viewed approximately 2030 times since records began.
SyntheticPagesTM are for use exclusively by those with training and experience in synthetic chemistry. While contributors are asked to identify hazards and to use methods designed to reduce risk, no formal hazard or risk assessments are included, and these procedures must be conducted at one's own risk. SyntheticPages, its editors, owners and associates do not warrant or guarantee the safety of individuals using these procedures and hereby disclaim any liability for any injuries or damages claimed to have resulted from or related in any way to the procedures herein. Copyright ©2006 Synthetic Pages Comment on this page!Registered users may add comments to other pages. If you have already registered, please log in, otherwise you may register here. |
|
| By using this site, you agree to its Terms and Conditions | ||







