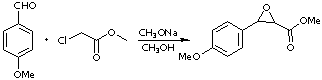Darzens condensation; Glycidic esters SyntheticPage 225 (2005)
Submitted 10th May 2005, published 11th May 2005
Kirtikumar B. Jadhav
([email protected]),
A contribution from the Chakraborti Group, NIPER
Chemicals Used
p-Methoxy benzaldehyde,
Methyl chloroacetate,
Methanol [distilled from Na],
Sodium metal
Procedure
To a solution of 5.1g (0.22 gram-atoms) of Na in 90mL of anhydrous MeOH, chilled to -10ºC in an ice bath, a solution of 20g (0.15 moles) of 4-Methoxy benzaldehyde and 23.9g (0.22 moles) of Methyl chloroacetate was added dropwise over a period of 3h, during which time the reaction mixture was vigorously stirred and finally the whole became a white paste. After the addition was completed, the mixture was stirred at -5ºC for 2h and then at room temperature for 3h. The mixture was poured into ice-water (350mL) containing AcOH (2mL); the precipitated white solid was filtered, washed with cold water and dried in a dessicator. The crude glycidate weighed 23g (75%). It was recrystallized from MeOH to give pure Methyl glycidate.
Author's Comments
It�s imperative to maintain inert (N2) atmosphere throughout the course of reaction. The glycidic ester is an important key intermediate in the synthesis of Diltiazem � a Ca2+ Channel blocker. The diastereomeric mixture obtained can further be purified by enzymatic methods in order to gain access to diltiazem.
Data
Melting range: 68-69ºC.
1H NMR (300MHz, CDCl3): 7.21 (2H, d, J = 8.6Hz), 6.89 (2H, d, J = 8.6Hz), 4.05 (1H, m*), 3.51 (1H, m*), 3.82 (3H, s), 3.80 (3H, s).
[* = as the reaction yields inseparable mixture of cis- and trans-glycidic ester, well resolved doublets for epoxide CH protons were not obtained].
APCI MS: 208.9 (M+1).
FT-IR (KBr, thin plate, ): 3020.2 cm-1 (w, sp2 C-H str), 2954.4 cm-1 (w, asymm. sp3 C-H str), 2837.7 cm-1 (w, symm. sp3 C-H str), 1728.3 cm-1 (vs, ester C=O str), 1613.8 - 1437 cm-1 (m, aro. skeletal C=C vibrations), 1311.7 cm-1 (m), 1294.0 cm-1 (m, symm str of epoxide), 1251.5 cm-1 (s, asymm. Ar-O-C str), 1179.0 cm-1 (w), 1029.4 cm-1 (s, symm Ar-O-C str), 856.1 cm-1 (w), 840.4 cm-1 (m, para subst. oop bend), 800.3 cm-1 (w), 774.2 cm-1( w).
Lead Reference
1. Ban, Y.; Oishi, T. Chem. Pharm. Bull., 1958, 6, 574-576.
2. Crotti, P.; Ferretti, M; Macchia, F. J. Org. Chem. 1986, 51, 2759-2766.
Other References
1. Bachelor, F.; Bansal, R. �The Darzens Glycidic Ester Condensation� J. Org. Chem. 1969, 34, 3600-3604.
2. Tung, C.; Speziale, A.; Frazier, H. �The darzens condensation. II. Reaction of chloroacetamides with aromatic aldehydes.� J. Org. Chem. 1963, 28, 1514-1521.
This page has been viewed approximately 1529 times since records began.
Get CDX file
SyntheticPagesTM are for use
exclusively by those with training and experience in synthetic chemistry. While contributors
are asked to identify hazards and to use methods designed to reduce risk, no formal hazard
or risk assessments are included, and these procedures must be conducted at one's own risk.
SyntheticPages, its editors, owners and associates do not warrant or guarantee the safety of
individuals using these procedures and hereby disclaim any liability for any injuries or damages
claimed to have resulted from or related in any way to the procedures herein. Copyright ©2005 Synthetic Pages
Comment on this page!
Registered users may add comments to other pages. If you have already registered, please
log in, otherwise you may
register here.
|